【Batch】Synthesis of [18F]-fluorination labeling reagent (Fluorination) 【バッチ】18-フッ素化標識試薬[18F]TDBFBの合成(フッ素化)
Tetrahedron Lett., 104, 154010 (2022).
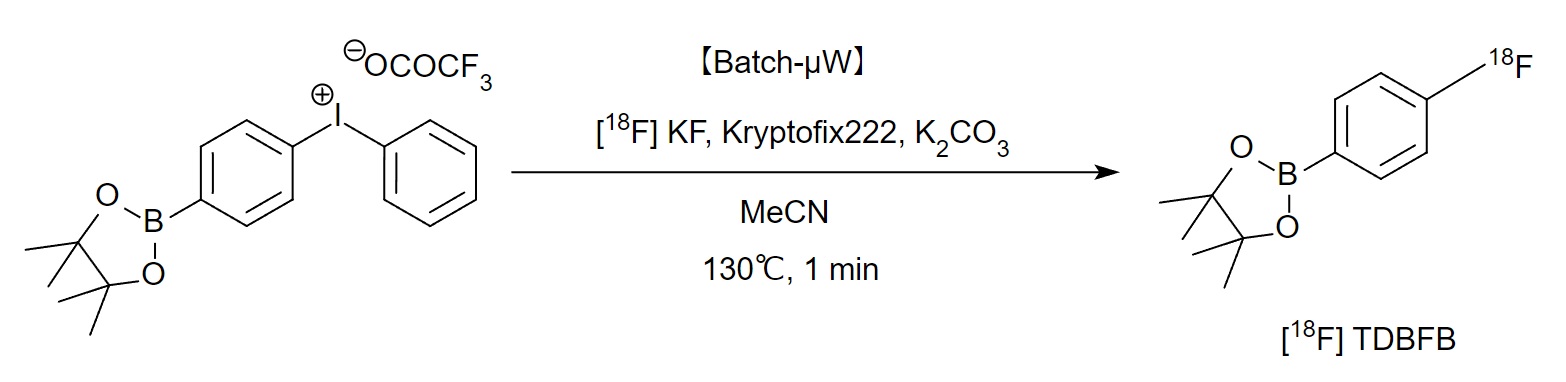
Outline
Tetrahedron Lett., 104, 154010 (2022).
DOI: 10.1016/j.tetlet.2022.154010
“Novel synthesis of an [18F]aryl boronic acid ester as a reagent for 18F-labeling via Suzuki coupling”
Yusuke Yagi a,b,c,d,⇑ , Hiroyuki Kimura b,⇑, Yuto Kondo b, Takahiro Higuchi c,d,e
a Department of Radiological Technology, Faculty of Medicinal Science, Kyoto College of Medical Science, 1-3 Imakita, Oyama-higashi, Sonobe, Nantan, Kyoto 622-0022, Japan
b Department of Analytical and Bioinorganic Chemistry, Division of Analytical and Physical Sciences, Kyoto Pharmaceutical University, 5 Nakauchi-cho, Misasagi, Yamashina-ku, Kyoto 607-8414, Japan
c Department of Nuclear Medicine, University Hospital Würzburg, OberdìrrbacherStrasse 6, 97080 Würzburg, Germany
d Comprehensive Heart Failure Center, University Hospital of Würzburg, Am Schwarzenberg 15, Haus A15, 97078 Würzburg, Germany
e Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, Okayama University, Okayama, Japan
Equipment information Reactor size: batch-type microwave reactor, Maximum power: 10 W (SAIDA FDS INC.)
概要
実験結果提供:
京都医療科学大学 屋木 祐亮先生
京都薬科大学 木村 寛之先生、近藤 悠斗様
ヴェルツブルク大学、岡山大学 樋口 隆弘先生
装置情報:バッチ型マイクロ波装置、10 W
